FDA Compliance. Simplified by AI.
FDA Compliance. Simplified by AI.
Komplyr analyzes labels, ingredients, and claims against FDA regulations in real time — preventing import holds, warning letters, and costly recalls.
Komplyr analyzes labels, ingredients, and claims against FDA regulations in real time — preventing import holds, warning letters, and costly recalls.




Built for Real-World FDA Compliance
Everything you need to detect risk, validate claims, and stay audit-ready — in one intelligent platform.

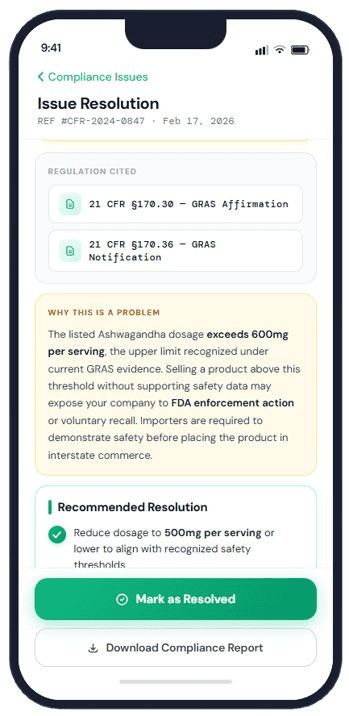
Regulation Citations & Documentation
Every flagged issue includes specific CFR references and plain-language explanations. Know exactly which regulation applies, and why, without decoding dense legal text.

Regulation Citations & Documentation
Every flagged issue includes specific CFR references and plain-language explanations. Know exactly which regulation applies, and why, without decoding dense legal text.
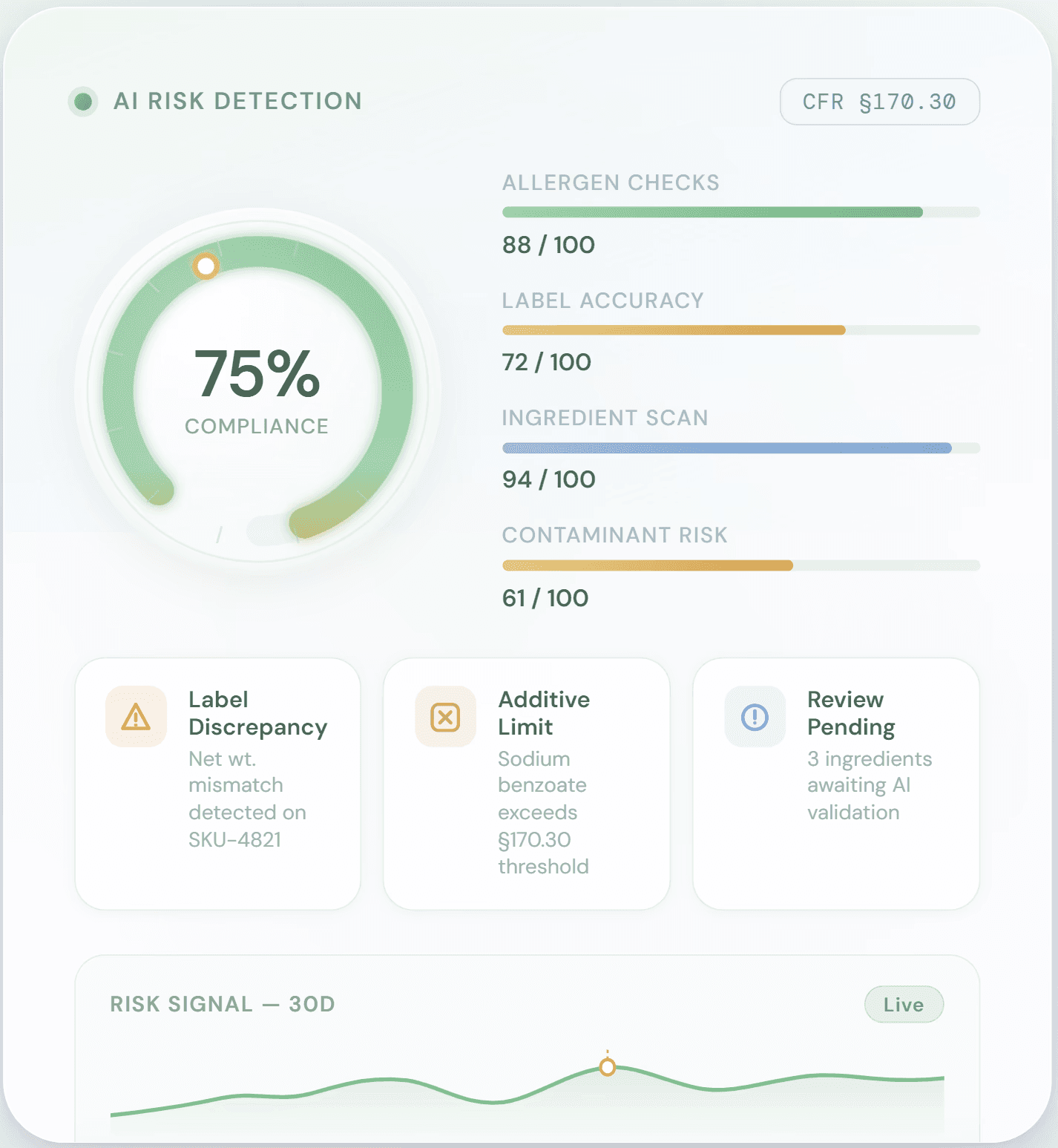
AI Risk Detection
Instantly analyze labels, ingredients, and claims against FDA regulations and CFR standards. Detect regulatory violations before they become warning letters, shipment holds, or costly recalls.

AI Risk Detection
Instantly analyze labels, ingredients, and claims against FDA regulations and CFR standards. Detect regulatory violations before they become warning letters, shipment holds, or costly recalls.
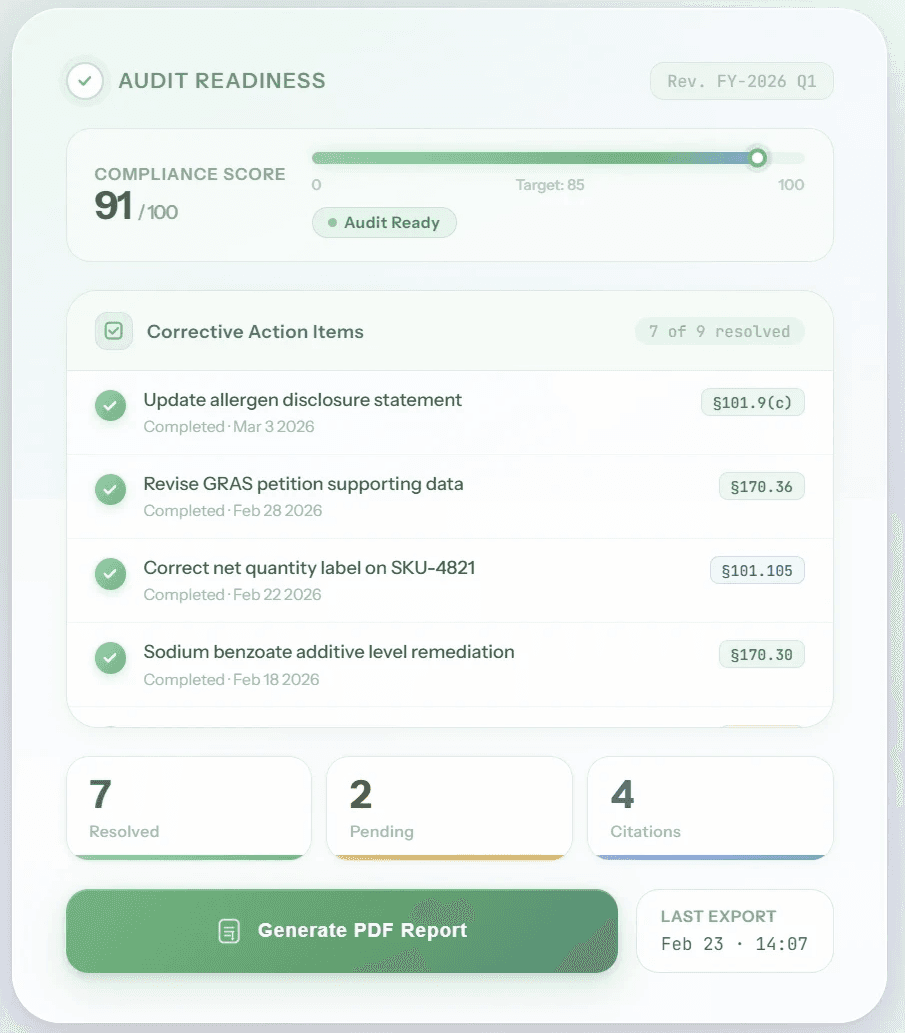
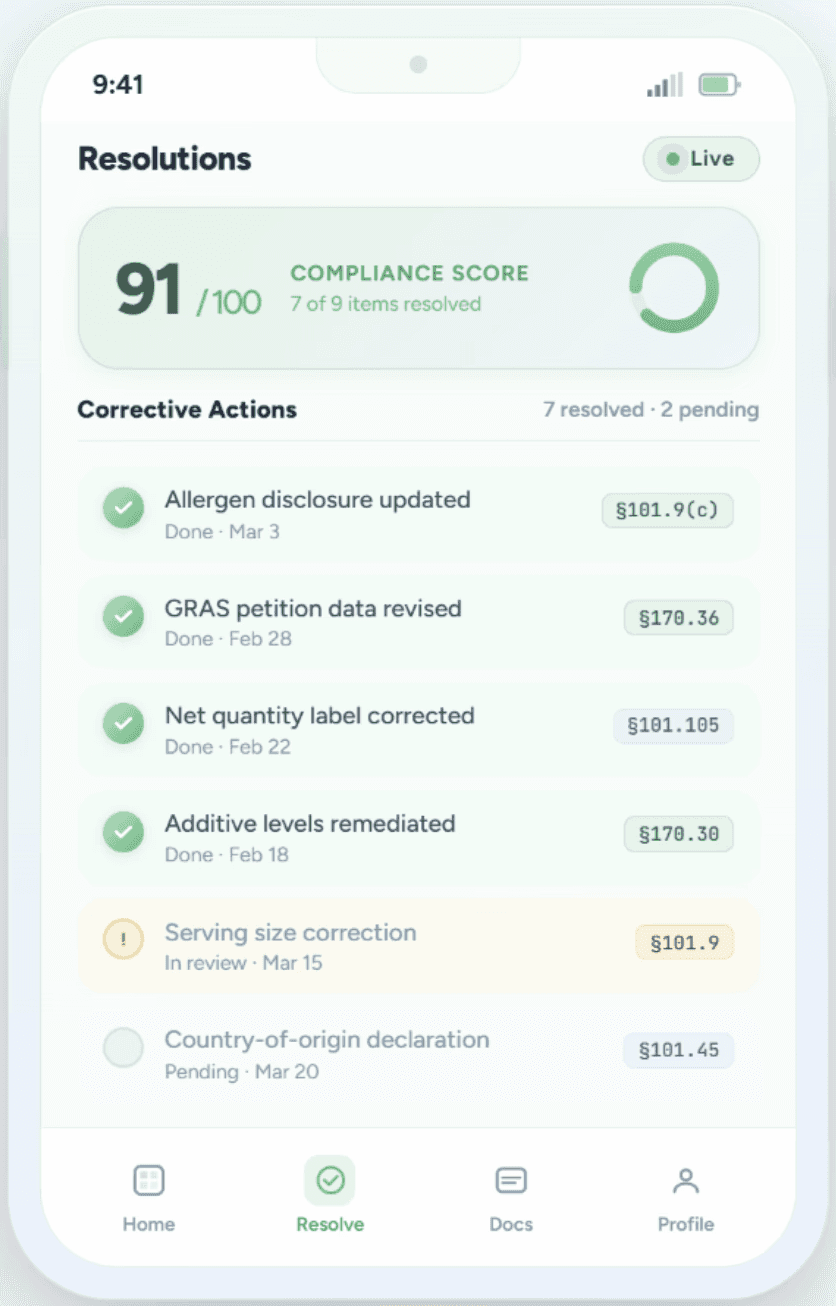
Resolution & Audit Readiness
Get step-by-step corrective recommendations and generate audit-ready compliance reports. Move from flagged issue to documented resolution in minutes, not weeks.

Resolution & Audit Readiness
Get step-by-step corrective recommendations and generate audit-ready compliance reports. Move from flagged issue to documented resolution in minutes, not weeks.
Built for Real-World FDA Compliance
Everything you need to detect risk, validate claims, and stay audit-ready — in one intelligent platform.

AI Risk Detection
Instantly analyze labels, ingredients, and claims against FDA regulations and CFR standards. Detect regulatory violations before they become warning letters, shipment holds, or costly recalls.

AI Risk Detection
Instantly analyze labels, ingredients, and claims against FDA regulations and CFR standards. Detect regulatory violations before they become warning letters, shipment holds, or costly recalls.

Regulation Citations & Documentation
Every flagged issue includes specific CFR references and plain-language explanations. Know exactly which regulation applies, and why, without decoding dense legal text.

Regulation Citations & Documentation
Every flagged issue includes specific CFR references and plain-language explanations. Know exactly which regulation applies, and why, without decoding dense legal text.

Resolution & Audit Readiness
Get step-by-step corrective recommendations and generate audit-ready compliance reports. Move from flagged issue to documented resolution in minutes, not weeks.

Resolution & Audit Readiness
Get step-by-step corrective recommendations and generate audit-ready compliance reports. Move from flagged issue to documented resolution in minutes, not weeks.


From Upload to Audit-Ready in Minutes
01
Upload Your Product Information
Submit your label, ingredient list, claims, or supplier documentation directly into the platform.
01
Upload Your Product Information
Submit your label, ingredient list, claims, or supplier documentation directly into the platform.
02
Identify Regulatory Risks
Instantly receive flagged issues with severity levels, CFR citations, and plain-language explanations.
02
Identify Regulatory Risks
Instantly receive flagged issues with severity levels, CFR citations, and plain-language explanations.
03
Resolve & Generate Documentation
Follow step-by-step corrective guidance and produce audit-ready compliance reports.
03
Resolve & Generate Documentation
Follow step-by-step corrective guidance and produce audit-ready compliance reports.


From Upload to Audit-Ready in Minutes
01
Upload Your Product Information
Submit your label, ingredient list, claims, or supplier documentation directly into the platform.
01
Upload Your Product Information
Submit your label, ingredient list, claims, or supplier documentation directly into the platform.
02
Identify Regulatory Risks
Instantly receive flagged issues with severity levels, CFR citations, and plain-language explanations.
02
Identify Regulatory Risks
Instantly receive flagged issues with severity levels, CFR citations, and plain-language explanations.
03
Resolve & Generate Documentation
Follow step-by-step corrective guidance and produce audit-ready compliance reports.
03
Resolve & Generate Documentation
Follow step-by-step corrective guidance and produce audit-ready compliance reports.
Enterprise-Grade Compliance Infrastructure



Secure, Cloud-Based Compliance
Access your regulatory reviews from anywhere — with encrypted storage and role-based access control.


Real-Time Regulatory Intelligence
Stay aligned with evolving FDA standards and CFR updates through continuously updated risk analysis models.
Audit-Ready Documentation
Generate structured compliance reports with embedded CFR citations.
Audit-Ready Documentation
Generate structured compliance reports with embedded CFR citations.
Automated Risk Scoring
Instant severity ratings for ingredient, label, and claim violations.
Automated Risk Scoring
Instant severity ratings for ingredient, label, and claim violations.
Import Risk Screening
Identify shipment-level compliance risk before products reach U.S. ports.
Import Risk Screening
Identify shipment-level compliance risk before products reach U.S. ports.
Enterprise-Grade Compliance Infrastructure




Secure, Cloud-Based Compliance
Access your regulatory reviews from anywhere — with encrypted storage and role-based access control.




Real-Time Regulatory Intelligence
Stay aligned with evolving FDA standards and CFR updates through continuously updated risk analysis models.
Audit-Ready Documentation
Generate structured compliance reports with embedded CFR citations.
Audit-Ready Documentation
Generate structured compliance reports with embedded CFR citations.
Automated Risk Scoring
Instant severity ratings for ingredient, label, and claim violations.
Automated Risk Scoring
Instant severity ratings for ingredient, label, and claim violations.
Import Risk Screening
Identify shipment-level compliance risk before products reach U.S. ports.
Import Risk Screening
Identify shipment-level compliance risk before products reach U.S. ports.

Built for Modern Food & Supplement Importers
Built for Modern Food & Supplement Importers

Designed with regulatory consultants and import professionals.

Designed with regulatory consultants and import professionals.

Designed with regulatory consultants and import professionals.



Compliance That Fits Every Regulatory Workflow
Compliance That Fits Every Regulatory Workflow
Whether you’re importing finished goods or launching a new supplement line, Komplyr adapts to your compliance needs.
Real-time label & ingredient analysis
Real-time label & ingredient analysis
Real-time label & ingredient analysis
App & Web developers
App & Web developers
App & Web developers
Supplement Brands
Supplement Brands
Supplement Brands
Marketing teams
Marketing teams
Marketing teams
Private Label Distributors
Private Label Distributors
Private Label Distributors
Multi-SKU E-commerce Brands
Multi-SKU E-commerce Brands
Multi-SKU E-commerce Brands
Know Your FDA Risk Before Your Next Shipment Ships.
Know Your FDA Risk Before Your Next Shipment Ships.
Know Your FDA Risk Before Your Next Shipment Ships.